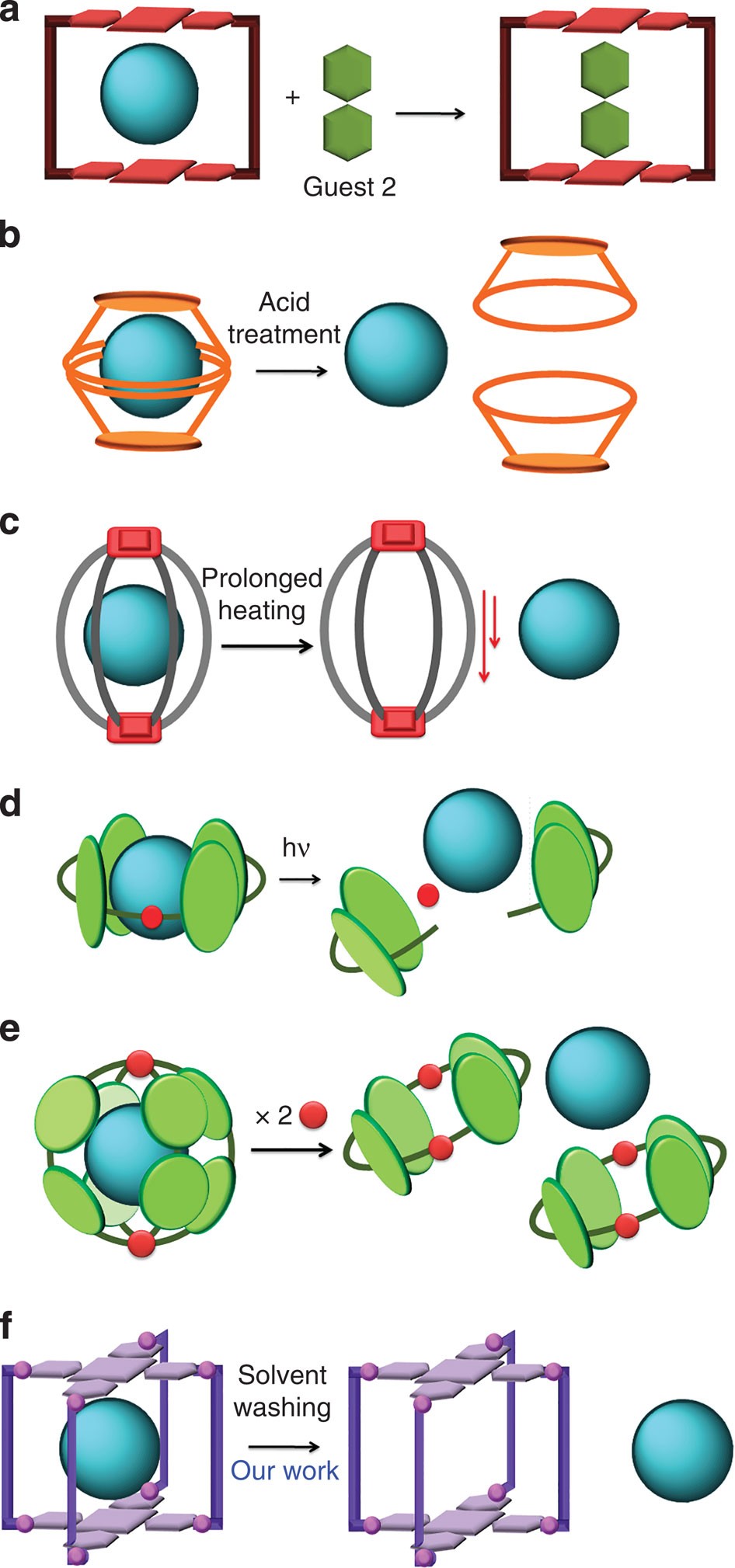
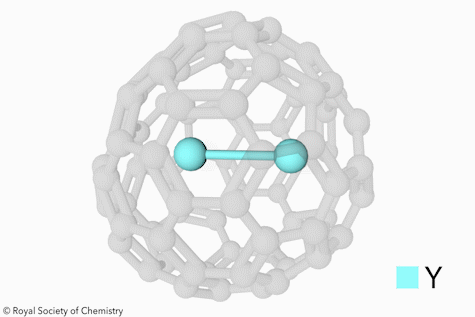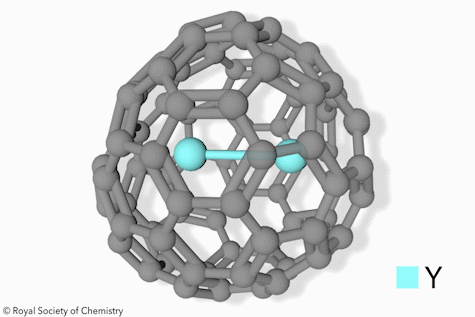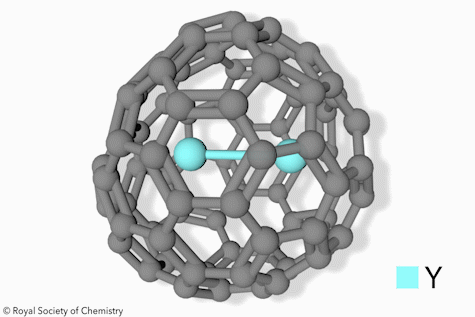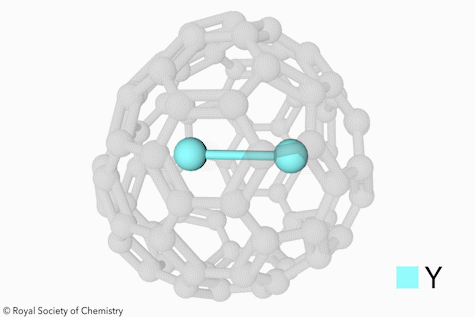Switchable Open‐Cage Fullerene for Water Encapsulation - Zhang - 2010 - Angewandte Chemie International Edition - Wiley Online Library

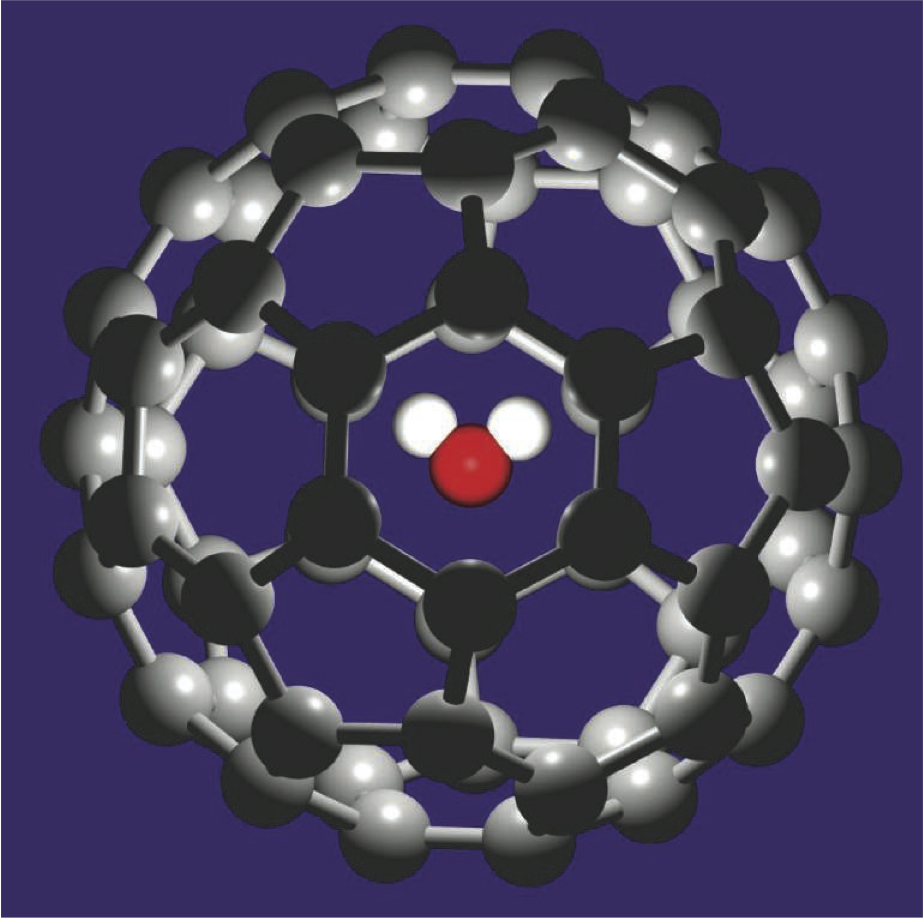
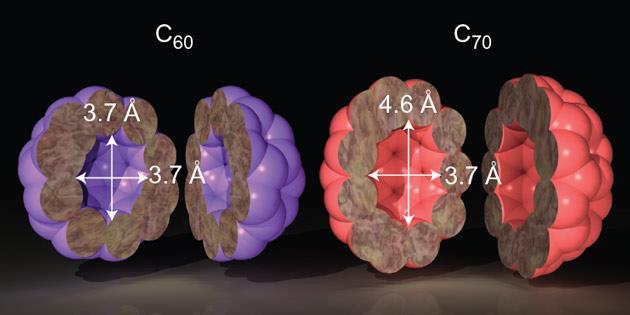
H2O@open-cage fullerene C60: control of the encapsulation property and the first mass spectroscopic identification - ScienceDirect

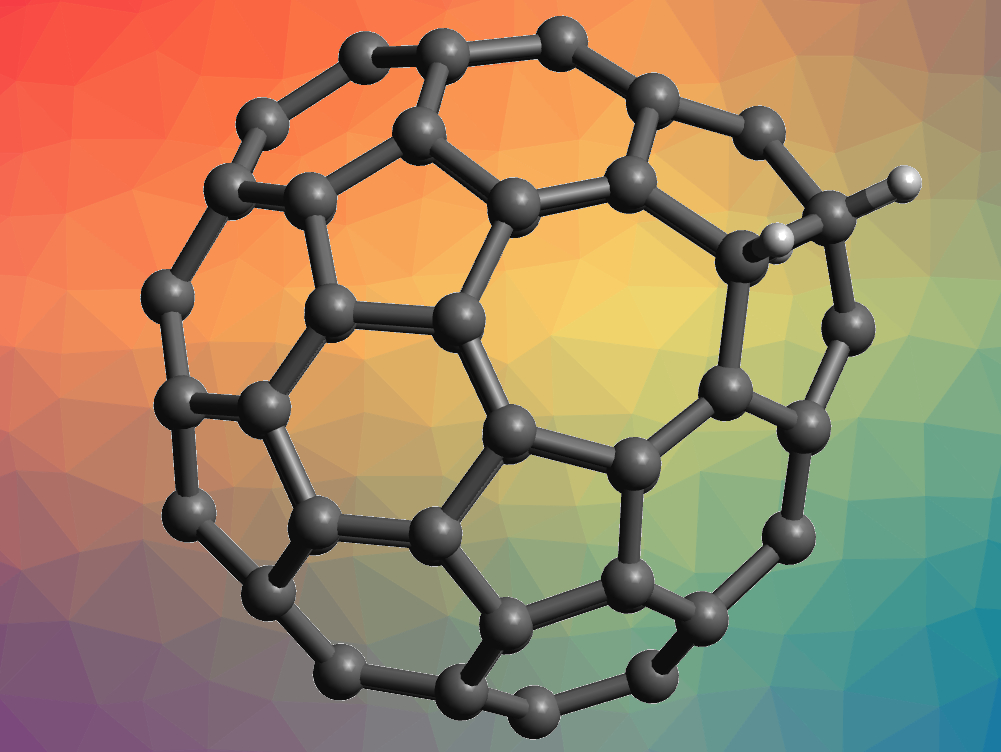
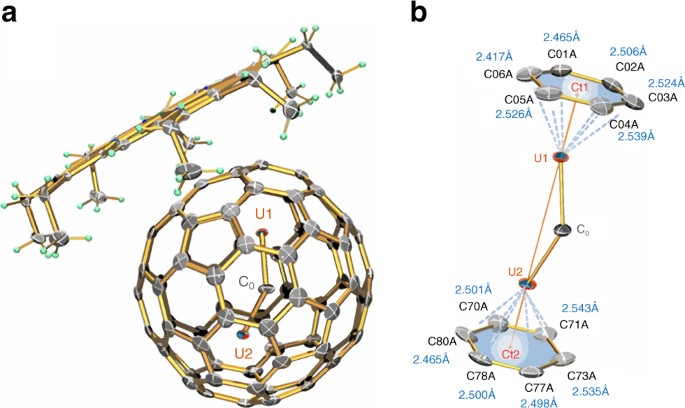
A Geometric Principle May Guide Self-Assembly of Fullerene Cages from Clathrin Triskelia and from Carbon Atoms: Biophysical Journal
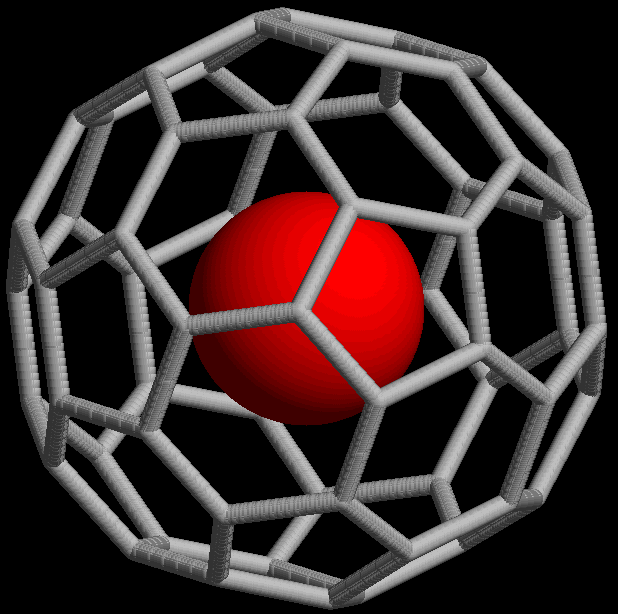
The stuffed-fullerene structure Stuff28-A). a Si 28 fullerene cage with... | Download Scientific Diagram
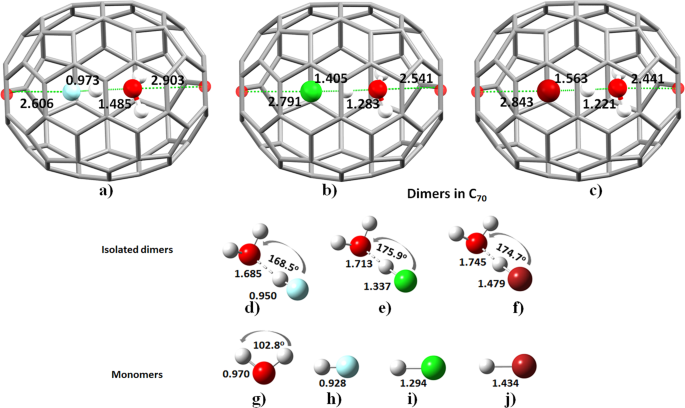
C70 Fullerene Cage as a Novel Catalyst for Efficient Proton Transfer Reactions between Small Molecules: A Theoretical study | Scientific Reports
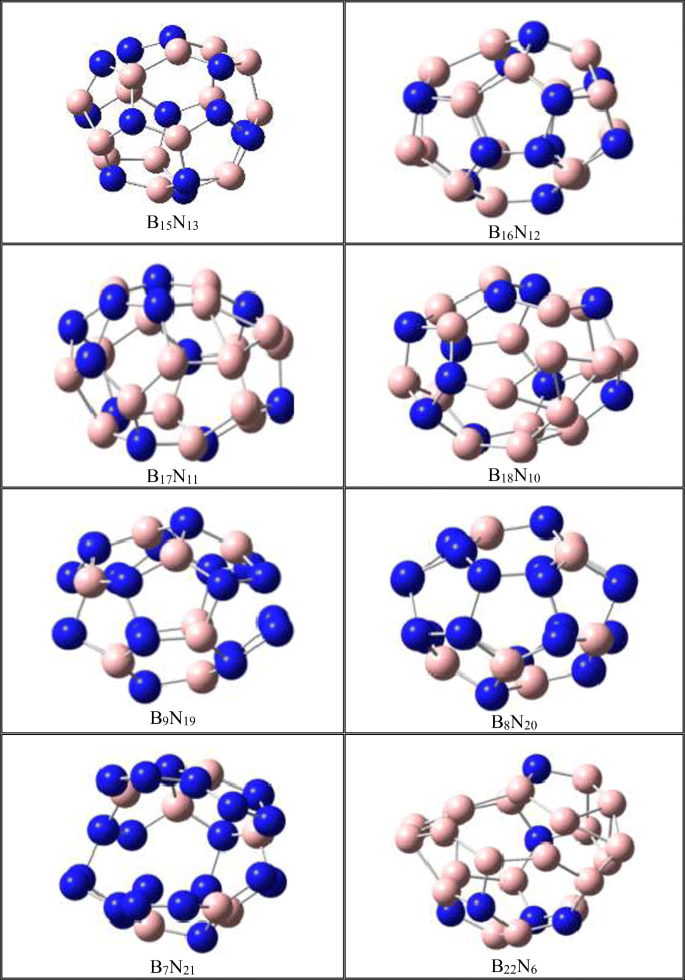
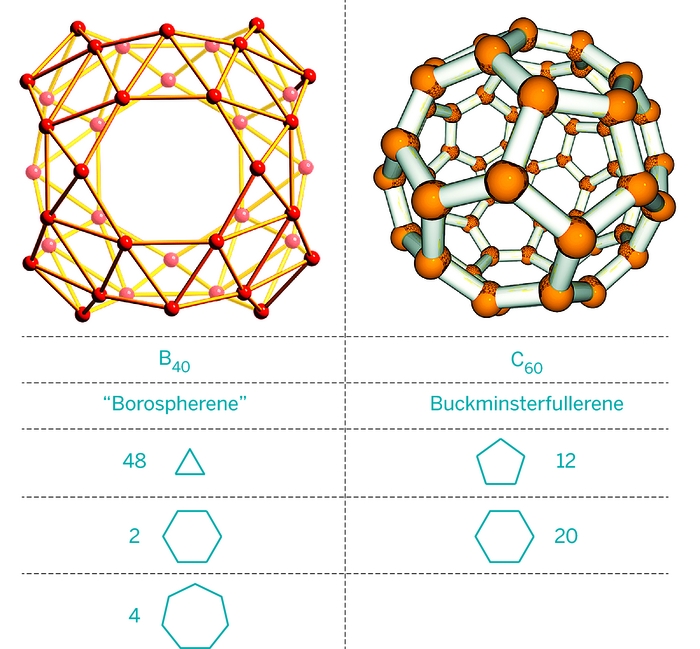
Fullerene-like boron nitride cages BxNy (x + y = 28): stabilities and electronic properties from density functional theory computation | SpringerLink

Open‐Cage Fullerene as Molecular Container for F−, Cl−, Br− and I− - Sun - Angewandte Chemie International Edition - Wiley Online Library
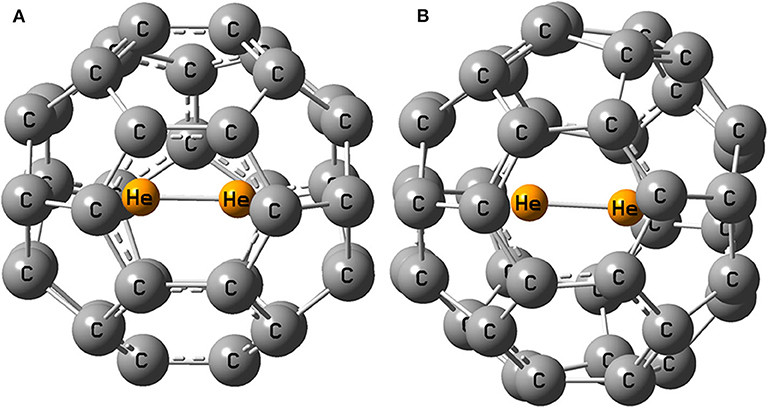
Frontiers | Confinement Effects of a Noble Gas Dimer Inside a Fullerene Cage: Can It Be Used as an Acceptor in a DSSC?

A geometric constraint, the head-to-tail exclusion rule, may be the basis for the isolated-pentagon rule in fullerenes with more than 60 vertices | PNAS